Nature has endowed human beings with many useful functions,
by virtue of supramolecular chemistry. As is well-known, nature
can self-assemble relatively small molecular precursors into very
complicated biomolecules. These supramolecular ensembles are
very important for various biological processes and entities such
as protein folding, phospholipid membranes, nucleic acid
assembly and tertiary structures, ribosomes, and microtubules.1
Supramolecular chemistry has been growing as a research area in
recent decades since the discovery of crown ethers, cryptands,
and spherands by three outstanding scientists, namely Pedersen,2
Lehn,3 and Cram,4 in the early 1960s; these achievements were
subsequently recognized by the awarding of the Nobel prize in
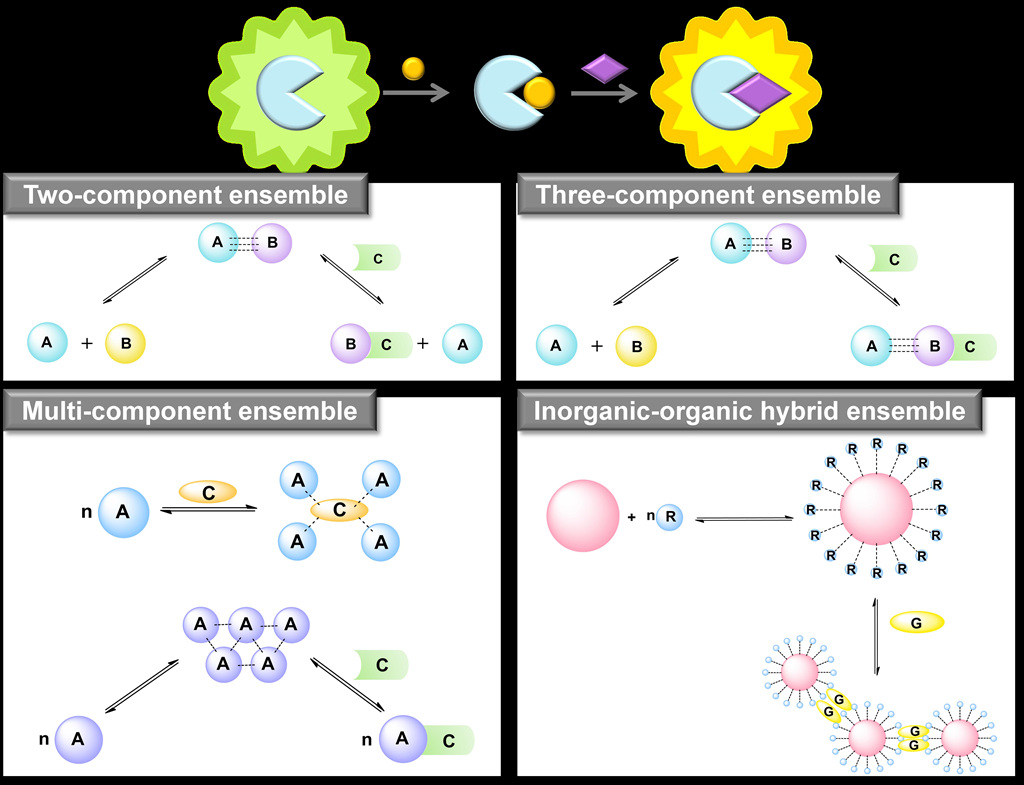
1987. Host−guest interactions, in which two or more
complementary molecules are able to recognize each other to
form a “programmed” supramolecular ensemble via natural
noncovalent interactions are known. Compared with the
precursor molecules, such preorganized supramolecular ensembles
have unusual optical, magnetic, sensing, and catalytic and
biological properties.5 Supramolecular chemistry has been
developed as a mature research field in modern science, and it
has interfaces with many other disciplines, including chemistry,
physics, materials, catalysis, and biology. It is a highly
interdisciplinary field that exceeds the conventional boundaries
of natural science. Supramolecular chemistry provides a good
opportunity for scientists to design new molecular materials with
various unusual structures and functions.
http://pubs.acs.org/doi/abs/10.1021/cr500553d