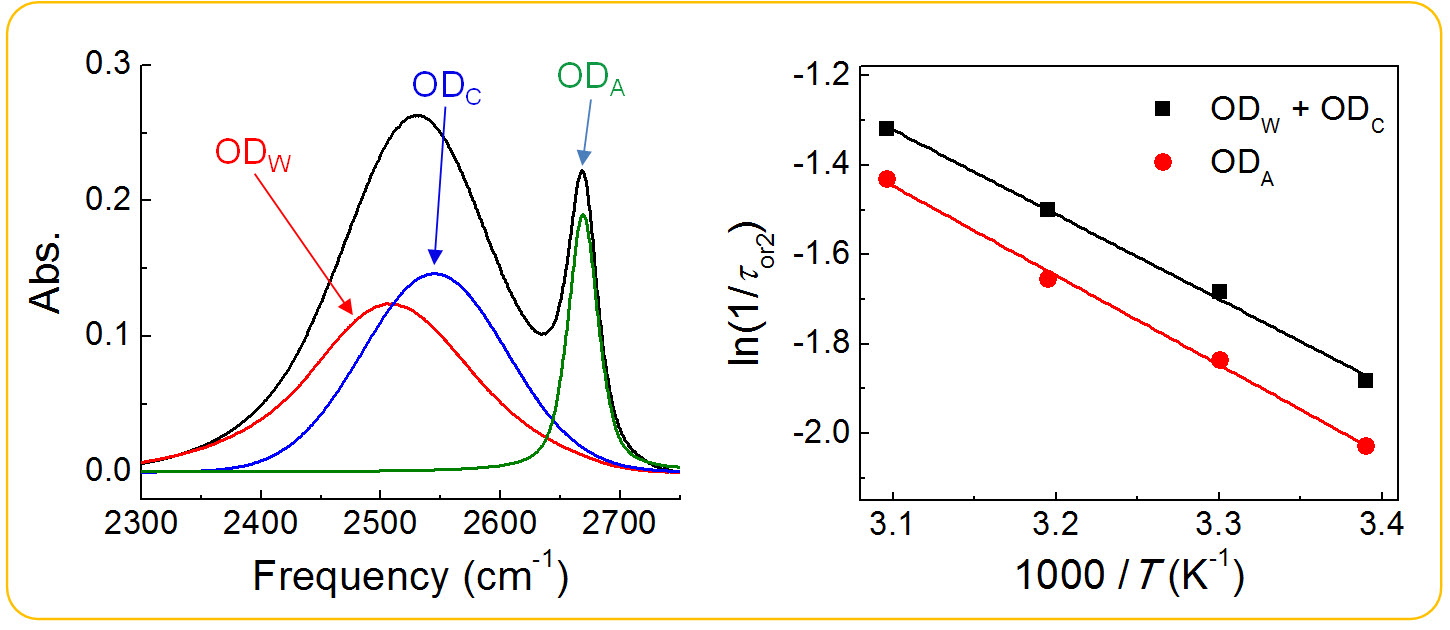
Dynamics of water in bulk and ionic hydration shells in aqueous ionic solutions are different because of the local environments. However, direct measurements of the dynamics of water in ionic hydration shells apart from those of bulk water are quite challenging experimentally because of poor spectral distinction between water molecules in bulk and ionic hydration shells. Interestingly, the hydroxyl stretch band in the FTIR spectrum of aqueous NaPF6 solution can be resolved into contributions from three distinct subsets of water: (1) water molecules hydrogen-bonded to other water (i.e. bulk water), (2) water molecules in the hydration shells of Na+ ions (i.e. cationic hydration shell), and (3) water molecules hydrogen-bonded to PF6
ions (i.e. anionic hydration shell). Such spectral features allowed us to study the individual dynamics of water in different subsets in aqueous NaPF6 solution. IR pump–probe spectroscopy was used to measure vibrational population relaxation, P(t), and orientational anisotropy decay, r(t), of water in different subsets. The vibrational lifetimes of water in cationic and anionic hydration shells in aqueous 5.0 M NaPF6 solution were directly determined and found to be independent of temperature up to 50 1C. Orientational anisotropy decay of water in ionic hydration shells was observed to be much slower than in bulk. r(t) became faster with increasing temperature, as predicted by the Debye–Stokes–Einstein equation. The activation energies for water orientation in different subsets were measured and found not to differ greatly in cationic and anionic hydration shells. These experiments allowed us to study the dynamics of water in bulk and ionic hydration shells in aqueous NaPF6 solutions in more detail.
http://pubs.rsc.org/en/Content/ArticleLanding/2014/CP/c4cp02823f#!divAbstract