Heejin Kim (교신저자)
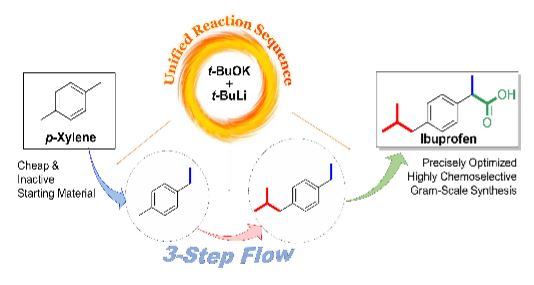
Ibuprofen was prepared from an inactive and inexpensive p-xylene by 3-step flow functionalizations through chemoselective metalations of benzyl positions in sequence using an in-situ generated LICKOR-type superbase. The flow approach in the microreactor facilitated to comprehensively explore over 100 conditions in the first-step reaction by varying concentrations, temperatures, solvents, and equivalents of reagents, enabling to find the optimal condition with 95% yield by significantly suppressing the formation of byproducts, followed by the second C–H metalation step in 95% yield. Moreover, gram-scale synthesis of ibuprofen in the final step was achieved by biphasic flow reaction of solution-phase intermediate with CO2, isolating 2.3 g for 10 min of operation time.
https://onlinelibrary.wiley.