Tae Su Choi(Post doc.) / Jong Yoon Han(박사과정)
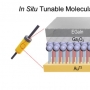
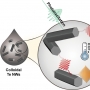
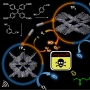
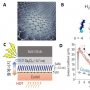
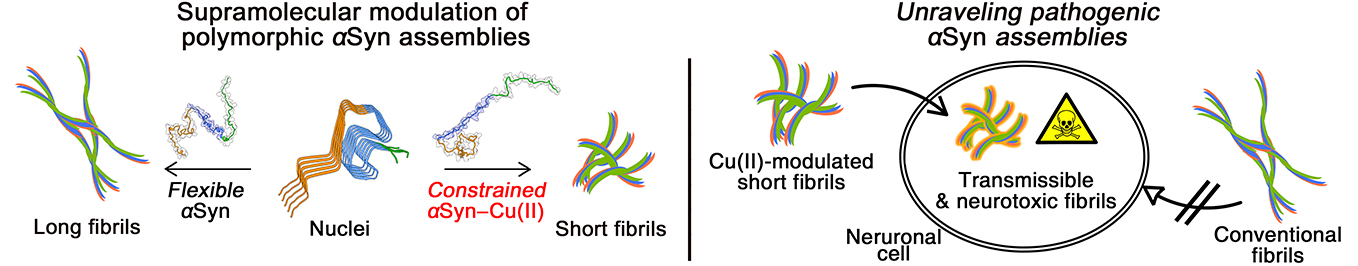
Structural variation of α-synuclein (αSyn) fibrils has been linked to the diverse etiologies of synucleinopathies. However, little is known about what specific mechanism provides αSyn fibrils with pathologic features. Herein, we demonstrate Cu(II)-based supra-molecular approach for unraveling the formation process of pathogenic αSyn fibrils and its application in a neurotoxic mechanism study. The conformation of αSyn monomer was strained by macrochelation with Cu(II), thereby disrupting the fibril elongation while promoting its nucleation. This non-canonical process formed shortened, β-sheet enriched αSyn fibrils (< 0.2 µm) that were rapidly transmitted and accumulated to neuronal cells, causing neuronal cell death, in sharp contrast to typical αSyn fibrils (~1 µm). Our approach provided the supramolecular basis for the formation of pathogenic fibrils through physiological factors such as brain Cu(II).
http://onlinelibrary.