“Organocatalytic Cascade Reactions for the Asymmetric Synthesis
of Fused-ring Compounds”
Fused-ring
structural units including chroman, indoline, pyrrolidine and hydroqunoline are
widely distributed in nature and many biologically active compounds. Molecules
containing these structural units exhibit a broad range of bioactivities such
as anticancer, antiviral, antitumar, antimicrobial, sex pheromone, and central
nervous system activity. Owing to the importance of these class of compounds, the stereoselective synthesis of fused-ring
compounds is a noteworthy synthetic
goal. In this presentation, we demonstrate the asymmetric synthesis of
hydroquinoline, hydroquinazoline, and indolines via catalytic cascade reaction. First,
A synthetic method for the
construction of fully substituted enantioenriched 1,4-dihydroquinolines using
an organocatalytic aza-Michael/Michael cascade reaction has been developed
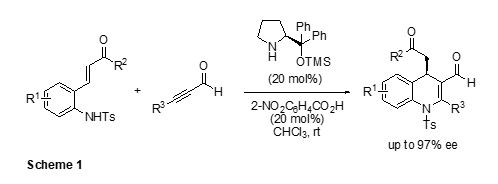
(Sheme 1). 1 The asymmetric reaction of 2-(tosylamino)phenyl
α,β-unsaturated ketones with alkynyl aldehydes, promoted by diphenylprolinol O-TMS ether as an organocatalyst,
generated chiral 1,4-dihydroquinolines in good to
high yields with excellent
enantioselectivities (up to 97% ee) We
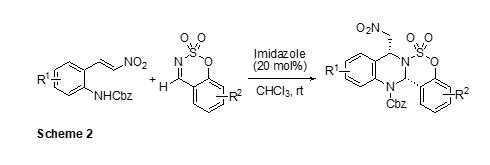
have also found a concise synthetic route to valuable highly functionalized
benzosulfamidate-fused tetrahydroquinazoline (Scheme 2).2 The [4+2]
cycloaddition of o-N-Cbz-amino-b-nitrostyrene with benzoxathiazine 2,2-dioxide using an imidazole as the
catalyst afforded tetrahydroquinazolines with high diastereoselectivities. In
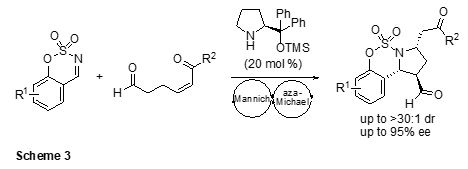
addition, we have established the synthesis of chiral benzosulfamidate-fused
pyrrolidines through an Mannich/aza-Michael cascade reaction of δ-formyl-a,b-unsaturated ketones with cyclic N-sulfimines
(Scheme 3).3 This
simple domino process afforded diverse highly functionalized pyrrolidines in good yields with high diastero- and
enantioselectivities. 1. Lee, Y.; Heo, S.; Kim, S.-G. Adv. Synth. Catal. 2015,
357, 1545-1550. 2. Sim, J.-T.; Kim, H. Kim, S.-G. Tetrahedron Lett. 2016, 57, 5907-5910. 3. Kim, H.; Kim, Y.; Kim, S.-G. J. Org. Chem. 2017, 82, 8179-8185.